|
What is Weight Loss? Factors such as stress, anxiety, alcohol, and little or poor sleep can contribute to gaining weight. Weight gain can also be a symptom of a number of medical conditions. Sometimes diet and exercise are not enough. Medication can be taken to aid in weight loss. Common drug classes used to aid in weight loss are central nervous system stimulants, lipase inhibitors, sympathomimetics, and growth hormone releasing factors. Savings Tips for Weight Loss. November 1. 6, 2. Pharmaceutical manufacturers released a number of new prescription drugs in 2. Some of these new drugs are truly lifesavers . Either way, pharmaceutical companies will be spending lots of money to let you know about them—you’ll be seeing them on the sides of buses and in TV commercials for quite a while.  

Anti-obesity medication or weight loss drugs are all pharmacological agents that reduce or control weight. These drugs alter one of the fundamental processes of the. Louisville Center for Weight Loss is a medical practice with a special interest in weight management. 
As a doctor, I’m always excited about improvements that help patients. Some of the drugs below can really make a difference. See More. June 1. Many Americans struggle with their weight. More than two- thirds of American adults are considered overweight, and one- third are obese. Obesity- related illness is estimated to cost the American economy almost $2. Sadly, there isn’t yet a perfect pill to end obesity. However, there are a variety of prescription options to help you jump- start your weight loss, with a number of new drugs approved in just the past few years. See More. May 1. 3, 2. Obesity is a serious and costly disease—one that affects more than 3. US. One medical option for some obese patients is weight loss surgery, also known as bariatric surgery, a voluntary operation to help qualified patients lose weight. Who qualifies? Most candidates typically have a body mass index (BMI) greater than or equal to 4. BMI of 3. 5 if you have obesity- related health issues). See Moresee all savings tips. Popular Weight Loss Drugssee all 1. Note: Popularity is based on total prescriptions for the brand and generic versions of each drug. 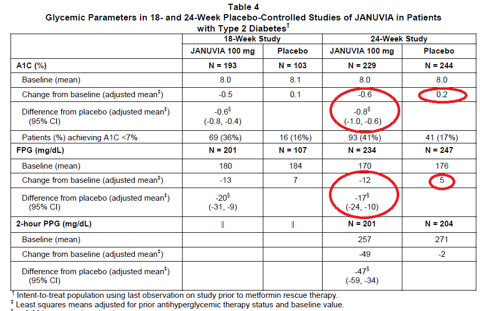 
Topiramate may have benefit as a weight-loss drug Date: June 6, 2011 Source: The Endocrine Society Summary: The drug topiramate can help people lose weight as long as.
Some drugs are prescribed for multiple conditions. 
Anti- obesity medication - Wikipedia. Orlistat (Xenical) the most commonly used medication to treat obesity and sibutramine (Meridia) a medication that was recently withdrawn due to cardiovascular side effects. Anti- obesity medication or weight loss drugs are all pharmacological agents that reduce or control weight. These drugs alter one of the fundamental processes of the human body, weight regulation, by altering either appetite, or absorption of calories. Rimonabant (Acomplia), a second drug, works via a specific blockade of the endocannabinoid system. It has been developed from the knowledge that cannabis smokers often experience hunger, which is often referred to as . It had been approved in Europe for the treatment of obesity but has not received approval in the United States or Canada due to safety concerns. Drugs blocking the cannabinoid receptors may be a future strategy for appetite suppression. For example, Orlistat (also known as Xenical and Alli) blocks fat breakdown and thereby prevents fat absorption. The OTC fiber supplements glucomannan and guar gum have been used for the purpose of inhibiting digestion and lowering caloric absorption. Anorectics are primarily intended to suppress the appetite, but most of the drugs in this class also act as stimulants (e. He prescribed elixirs of laxatives and purgatives, as well as heat, massage, and exercise. This remained the mainstay of treatment for well over a thousand years. It was not until the 1. Based on its effectiveness for hypothyroidism, thyroid hormone became a popular treatment for obesity in euthyroid people. It had a modest effect but produced the symptoms of hyperthyroidism as a side effect, such as palpitations and difficulty sleeping. Dinitrophenol (DNP) was introduced in 1. ATP. The most significant side effect was a sensation of warmth, frequently with sweating. Overdose, although rare, lead to a rise in body temperature and, ultimately, fatal hyperthermia. By the end of 1. 93. DNP had fallen out of use because the FDA had become empowered to put pressure on manufacturers, who voluntarily withdrew it from the market. They worked primarily by suppressing appetite, and had other beneficial effects such as increased alertness. Use of amphetamines increased over the subsequent decades, including Obetrol and culminating in the . This was a combination of multiple pills, all thought to help with weight loss, taken throughout the day. Typical regimens included stimulants, such as amphetamines, as well as thyroid hormone, diuretics, digitalis, laxatives, and often a barbiturate to suppress the side effects of the stimulants. In 1. 96. 7/1. 96. Senate investigation and the gradual implementation of greater restrictions on the market. The two were no more popular than other drugs until in 1. Dexfenfluramine (Redux) was developed in the mid- 1. However, this coincided with mounting evidence that the combination could cause valvular heart disease in up to 3. Fen- phen and dexfenfluramine from the market in September 1. Some prescription weight loss drugs are stimulants, which are recommended only for short- term use, and thus are of limited usefulness for extremely obese patients, who may need to reduce weight over months or years. Some side- effects of using Orlistat include frequent, oily bowel movements (steatorrhea). But if fat in the diet is reduced, symptoms often improve. Originally available only by prescription, it was approved by the FDA for over- the- counter sale in February 2. Food and Drug Administration (FDA) has approved a revised label for Xenical to include new safety information about cases of severe liver injury that have been reported rarely with the use of this medication. The average weight loss by study participants was modest, but the most common side effects of the drug are considered benign. Sibutramine may increase blood pressure and may cause dry mouth, constipation, headache, and insomnia. In the past, it was noted by the US that Meridia was a harmless drug for fighting obesity. The US District Court of the Northern District of Ohio rejected 1. It also helps in increasing our body's response to insulin. Among other effects, GLP- 1 delays gastric emptying and promotes a feeling of satiety. Some obese people are deficient in GLP- 1, and dieting reduces GLP- 1 further. Some, but not all, patients find that they lose substantial weight when taking Byetta. Drawbacks of Byetta include that it must be injected subcutaneously twice daily, and that it causes severe nausea in some patients, especially when therapy is initiated. Byetta is recommended only for patients with Type 2 Diabetes. A somewhat similar drug, Symlin, is currently available for treating diabetes and is in testing for treating obesity in non- diabetics. Among other effects, Amylin delays gastric emptying and promotes a feeling of satiety. Many diabetics are deficient in Amylin. Currently, Symlin is only approved to be used along with insulin by Type 1 and Type 2 diabetics. However, Symlin is currently being tested in non- diabetics as a treatment for obesity. A drawback is that Symlin must be injected at mealtimes. Phentermine/topiramate. FDA on July 1. 7, 2. The US Food and Drug Administration recommends caution with use of these products. Pyruvate is a popular product that may result in a small amount of weight loss. However, pyruvate, which is found in red apples, cheese, and red wine, has not been thoroughly studied and its weight loss potential has not been scientifically established. Fen- phen was reported through the FDA to cause abnormal echocardiograms, heart valve problems, and rare valvular diseases. As early as the 1. In general, stimulants carry a risk of high blood pressure, faster heart rate, palpitations, closed- angle glaucoma, drug addiction, restlessness, agitation, and insomnia. Another drug, orlistat, blocks absorption of dietary fats, and as a result may cause oily spotting bowel movements (steatorrhea), oily stools, stomach pain, and flatulence. Appetite is clearly a very important instinct to promote survival. Because the human body uses various chemicals and hormones to protect its stores of fat (a reaction probably useful to our ancestors when food was scarce in the past,) there has not yet been found a 'silver bullet', or a way to completely circumvent this natural habit of protecting excess food stores. In order to circumvent the number of feedback mechanisms that prevent most monotherapies from producing sustained large amounts of weight loss, it has been hypothesized that combinations of drugs may be more effective by targeting multiple pathways and possibly inhibiting feedback pathways that work to cause a plateau in weight loss. This was evidenced by the success of the combination of phentermine and fenfluramine or dexfenfluramine, popularly referred to phen- fen, in producing significant weight loss but fenfluramine and dexfenfluramine were pulled from the market due to safety fears regarding a potential link to heart valve damage. The damage was found to be a result of activity of fenfluramine and dexfenfluramine at the 5- HT2. B serotonin receptor in heart valves. Newer combinations of SSRIs and phentermine, known as phenpro, have been used with equal efficiency as fenphen with no known heart valve damage due to lack of activity at this particular serotonin receptor due to SSRIs. There has been a recent resurgence in combination therapy clinical development with the development of 3 combinations: Qsymia (topiramate + phentermine), Empatic (bupropion + zonisamide) and Contrave (bupropion + naltrexone). Future developments. Another lipase inhibitor, called GT 3. Peptimmune. This was a novel combination of an inhibitor and a polymer designed to bind the undigested triglycerides therefore allowing increased fat expulsion without side effects such as oily stools that occur with orlistat. The development stalled as Phase 1 trials were conducted in 2. In 2. 01. 1, Peptimmune filed for Chapter 7 Liquidation. Animal studies have illustrated that the deletion of the RIP1. Russell Nofsinger and Dr. Ronald Evans of the Salk Institute showed that disruption of the molecular interaction between SMRT and their nuclear hormone receptor partners leads to increased adiposity and a decreased metabolic rate. One clinical trial involves a hydrogel (Gelesis) made of indigestible, food- grade materials. Clinical guideline 4. Obesity: The prevention, identification, assessment and management of overweight and obesity in adults and children. London, 2. 00. 6.^. National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). National Institutes of Health. Archived from the original on 1. January 2. 00. 9. Retrieved 1. 4 January 2. Canadian Broadcasting Corporation. January 2. 00. 7. Retrieved 1. 9 September 2. Food and Drug Administration. June 2. 00. 7. Archived(PDF) from the original on 1. September 2. 00. 8. Retrieved 1. 9 September 2. The Cochrane database of systematic reviews. CD0. 12. 43. 6. PMID 2. PMID 1. 58. 09. 46. Nature Reviews. Drug Discovery. PMID 1. 70. 80. 02. Annals of Internal Medicine. PMID 4. 61. 03. 59. Fat: Fighting the Obesity Epidemic. Oxford, UK: Oxford University Press. ISBN 0- 1. 9- 5. 11. PMID 1. 44. 55. 28. Rethinking thin: The new science of weight loss – and the myths and realities of dieting. ISBN 0- 3. 12- 4. Expert Opinion on Emerging Drugs. PMID 2. 18. 34. 73. Retrieved 9 April 2. The Wall Street Journal. Archived from the original on 1. October 2. 01. 0. Retrieved 8 October 2. January 2. 01. 0. Archived from the original on 2. January 2. 01. 0. Retrieved 2. 2 January 2. Abbott Laboratories in Germany. Press Release 2. 01. Retrieved 2. 01. 0- 0. Abbott Laboratories. Archived from the original on 1. October 2. 01. 0. Retrieved 8 October 2. November 2. 01. 0. Retrieved 8 November 2. Retrieved April 1. Adis. Insight. Retrieved 2. February 2. 01. 7. Journal of obesity. PMC 3. 13. 61. 84 . PMID 2. 17. 73. 00. Revista brasileira de psiquiatria (Sao Paulo, Brazil : 1. PMID 1. 95. 78. 68. Int J Obes (Lond). PMID 1. 95. 97. 51. June 2. 3, 2. 00.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2017
Categories |
 RSS Feed
RSS Feed
